Antibiotic Resistance FHIR Format Examples Now Available!
The World Health Organization (WHO) has designated antibiotic resistance as one of the ten greatest global health threats. Effectively recording, integrating, and utilizing antimicrobial resistance data has become an urgent priority in clinical medicine and scientific research! Dr. Hung-Yi Chiou, Director of the Institute of Population Health Sciences at the National Health Research Institutes; Professor Hsiao-Hui Tsou; and Assistant Chieh Cheng collaborated with infectious disease expert Dr. Shu-Chen Kuo; FHIR format experts Professor Yi-Hsin Yang; Dr. Hsiu-An Lee, and Associate Professor Li-Hui Lee (Figure 1) to develop a Taiwan-specific antibiotic resistance data format template. This template helps systematize and standardize data previously scattered across various stages of the testing process. It not only enhances the accuracy of data recording but also facilitates data exchange between hospitals, further expanding its application value for clinical decision-making and research analysis!
Figure 1: Contributors
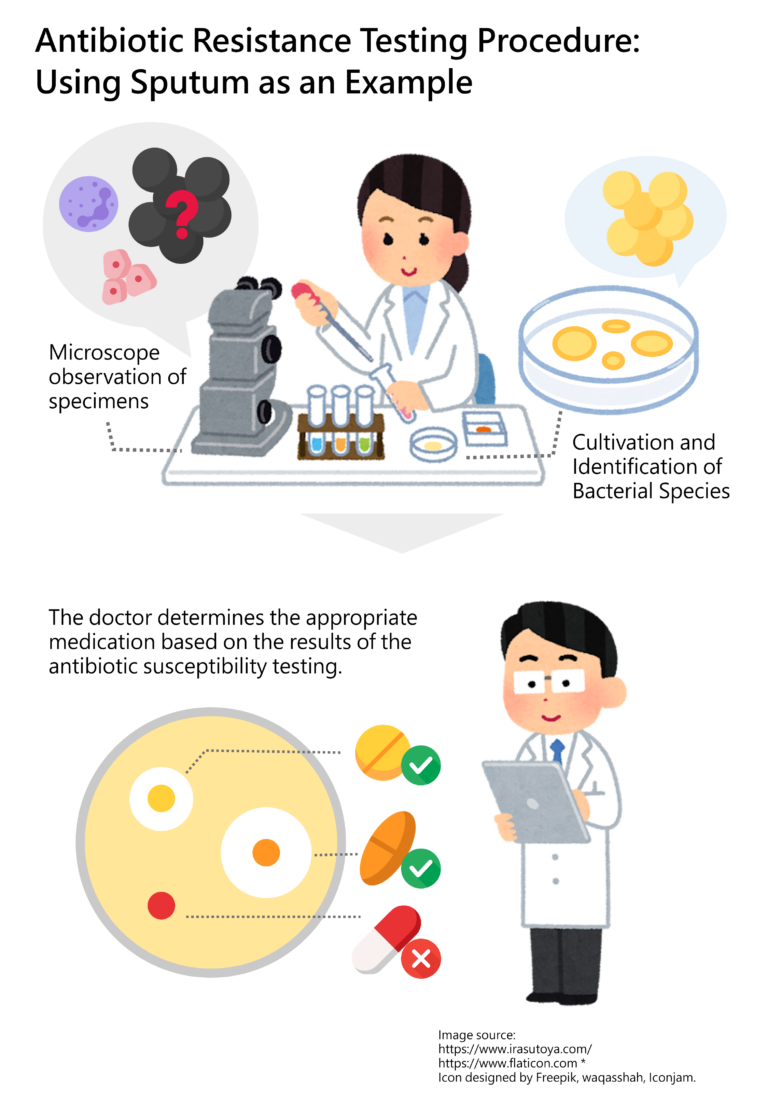
If a bacterial infection occurs, how can we determine which antibiotic is effective? In hospitals, a comprehensive antibiotic test involves several crucial steps. Taking the assessment of bacterial resistance in sputum as an example, the process begins with a microscopic examination. This initial step involves observing the specimen (sputum) for the presence of cells (such as epithelial cells), white blood cells, or bacteria. While bacterial characteristics may be visible at this stage, the specific species cannot yet be confirmed. Therefore, the bacteria in the sample are simultaneously cultured and identified. Only after confirming the bacterial species can further testing be conducted to assess its response to various antibiotics—this is the antibiotic susceptibility testing. Based on these results, the doctor selects the most effective and appropriate antibiotic for the patient’s treatment. (Figure 2)
Figure 2: Antibiotic Resistance Testing Procedure
Clearly documenting each interconnected testing step not only ensures medical accuracy but also enables rapid understanding by other hospitals during referrals. Furthermore, accumulating this data into a long-term database to observe overall trends in antibiotic resistance can provide immense benefits to both patients and society! Therefore, the team adopted the internationally recognized FHIR (Fast Healthcare Interoperability Resources) format as the standard and followed the design specifications outlined in the CDM IG (Common Data Model Implementation Guide) to structure the data.
This example provides simulated antibiotic resistance test reports, architectural diagrams, JSON files, and SNOMED CT (Systematized Nomenclature of Medicine — Clinical Terms) code tables for common pathogens. It aims to establish a solid foundation for clinical healthcare and research applications.
For related materials, please refer to the “Taiwan Infectious Disease Thematic Database: FHIR Zone” managed by the Institute of Population Health Sciences, National Health Research Institutes.
抗生素抗藥性FHIR格式範例上線!
人工智慧(AI)的快速發展,為醫療領域帶來了新的可能性。面對龐大而複雜的抗生素抗藥性資料,除了仰賴人類專業判讀,要如何讓電腦也看得懂,成為得力的助手?國家衛生研究院群體健康科學研究所邱弘毅所長、鄒小蕙老師及助理成捷攜手感染症專家郭書辰醫師、FHIR格式專家楊奕馨老師、李修安博士與李麗惠老師共同協作(圖一),打造適合台灣的抗生素抗藥性資料格式範例,範例可協助將原本分散於檢驗流程各階段的資料系統化、標準化,不僅提升資料紀錄的精準度,也有助於醫院間的資料交換,進一步擴大臨床決策與研究分析的應用價值!
圖一、範例貢獻者
回顧新冠疫情,微小的病原體在短時間內席捲全球,帶來難以估計的生命損失與社會衝擊,即使疫情遠去仍令人心有餘悸。然而,在我們看不見的角落,另一場更無聲、卻同樣危險的威脅正悄然累積——抗生素抗藥性。當細菌逐漸對原本有效的藥物產生抵抗能力,造成藥物效果變差或是沒有作用,「無藥可醫」將不再只是警告,而可能成為醫療現場的日常。正因如此,世界衛生組織(WHO)已將抗生素抗藥性列為全球十大健康威脅之一。如何有效紀錄、整合並善用抗藥性資料,已成為臨床醫療與科學研究中刻不容緩的關鍵課題。
假如細菌感染,要如何知道哪一種抗生素有效?在醫院,一次完整的抗生素檢驗包含多個重要步驟。以檢驗痰液中細菌的抗藥性為例,首先會進行顯微鏡檢查,初步觀察檢體(痰液)中是否有細胞 (如上皮細胞)、白血球或細菌等等,此時雖然能觀察到細菌的特徵,但還不能確認種類,因此同時會將檢體中的細菌進行培養與鑑定,在確認細菌種類後,才能進一步測試它對各種抗生素的反應,也就是抗藥性檢測。醫師會根據這些結果,選擇對病患最有效、最合適的抗生素進行治療。(圖二)
圖片來源: https://www.irasutoya.com/ https://www.flaticon.com * Icon designed by Freepik, waqasshah, Iconjam.
圖二、抗生素抗藥性檢驗檢查流程
這些檢驗步驟環環相扣,每一個結果都需要被清楚、正確地記錄下來,才能確保後續醫療決策的正確性。更進一步來說,若這些資料能在病患轉診時,讓其他醫院快速理解,或是長期累積成資料庫,用來觀察整體抗生素抗藥性的變化趨勢,將能對病患以及社會帶來更大的幫助。
要達成這些目標,統一資料格式就顯得格外重要。團隊以國際通用的 FHIR(Fast Healthcare Interoperability Resources)格式作為標準,並依循 CDM IG (Common Data Model Implementation Guide) 的設計規範來撰寫資料。CDM IG不僅符合台灣核心規範(TW Core IG),設計時也以檢驗檢查資料檔案為原型,更貼近醫院現有的常用格式。
本範例提供模擬的抗生素抗藥性檢驗檢查報告、架構圖、JSON檔案以及常用病原體的SNOMED CT(Systematized Nomenclature of Medicine — Clinical Terms)代碼表。檢驗報告內容涵蓋顯微鏡檢查、細菌培養與鑑定,以及抗生素抗藥性檢測三大部分;架構圖幫助讀者理解檢驗資料轉換成 FHIR 格式的結構;驗證成功的 JSON 檔案,呈現最詳細、完整的資料內容;最後,SNOMED CT病原體代碼表完整包含了常見細菌,減少查找代碼的時間。所有範例皆經感染症專家與 FHIR 專家審核,將複雜的細菌與抗生素抗藥性資訊,以更清楚、易懂的方式呈現,為臨床醫療與研究應用打下良好的基礎。
最後更新時間:2026/3/19
參考資料:
台灣核心實作指引: https://twcore.mohw.gov.tw/ig/twcore/index.html
國家級人體生物資料庫整合平台共同資料模式實作指引:https://www.ghd.tw/cdm-fhir/index.html
NHS England Pathology FHIR Implementation Guide: https://simplifier.net/guide/pathology-fhir-implementation-guide/Home/FHIRAssets/AllAssets/All-Profiles/Examples/Bundles/Urine-MC-S-Report-Structured?version=0.1
HL7 Europe Laboratory Report: https://hl7.eu/fhir/laboratory/Bundle-BundleLabResultMicroCultureSusc.html#Observation_1097929f-890e-4e27-a31d-58599f3e2479
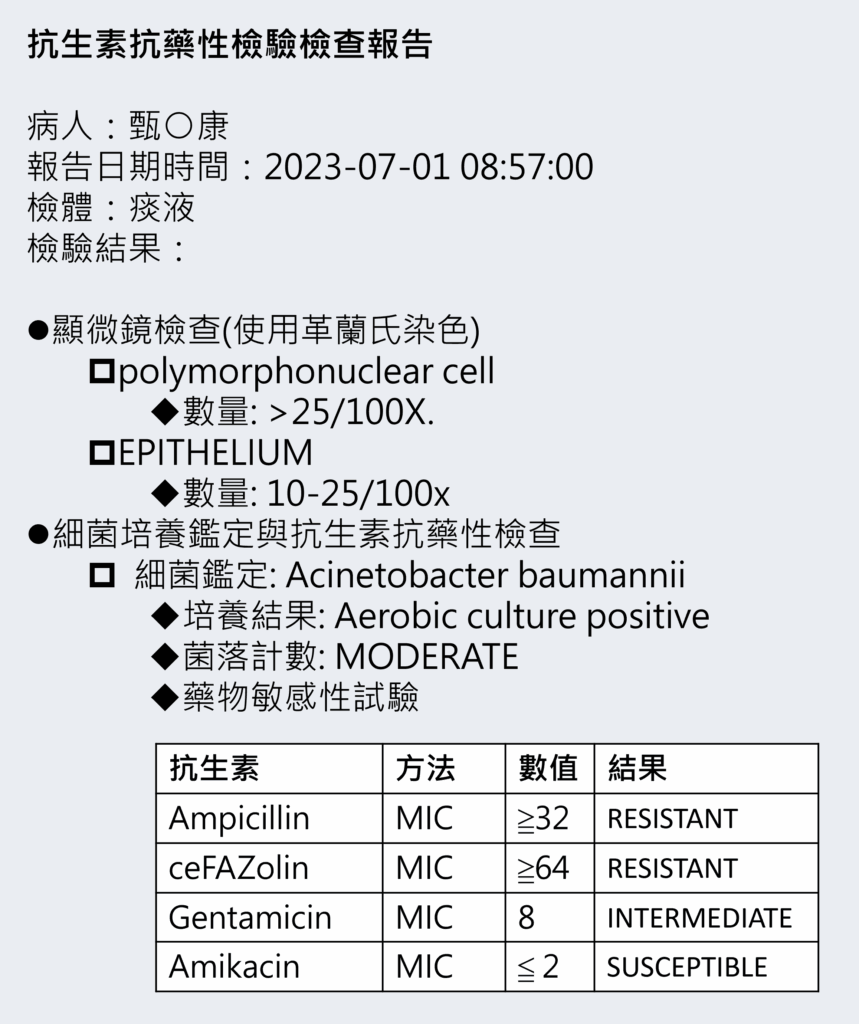
此為模擬檢驗檢查的相關單張,目的為說明抗生素抗藥性檢驗檢查預期會進行的流程與結果。
此圖為簡化之資料架構示意圖,為使範例清楚易懂,僅呈現主要資源之間的核心關聯,未涵蓋所有關聯性。如需查閱各 Resource 之完整關聯與詳細定義,請參閱 Common Data Model Implementation Guide (IG)。此外,資料架構中之 Patient、Encounter、Specimen 與 Organization 等 Resource,該IG 已有完整說明與範例。為避免內容重複,本文不再另行列出,請依實際需求參考該 IG 進行調整與實作。
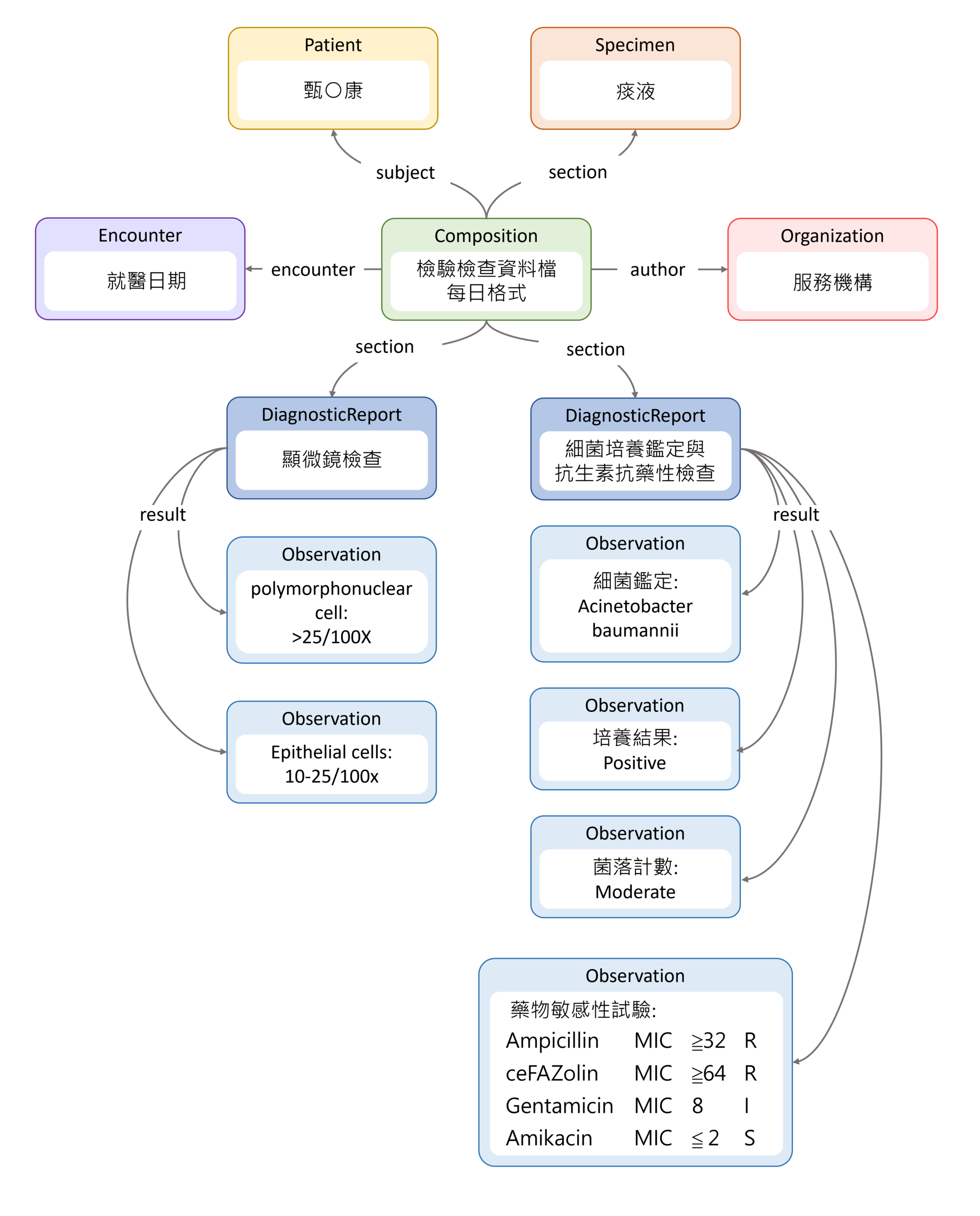
本範例撰寫感謝鄒小蕙老師、郭書辰醫師、楊奕馨老師、李修安博士、李麗惠老師與助理成捷共同協作。
{ "resourceType": "Composition", "id": "com", "status": "final", "type": { "coding": [{ "system": "https://www.ghd.tw/cdm-fhir/CodeSystem/ReportType", "code": "1", "display": "檢體檢驗報告" }] }, "subject": { "reference": "Patient/pat-CDM" }, "encounter": { "reference": "Encounter/enc-LABD" }, "date": "2023-07-01T08:57:00+08:00", "author": [{ "reference": "Organization/org-LABDM" }], "title": "檢驗檢查資料檔每日格式 LABD", "section": [{ "code": { "coding": [{ "system": "http://loinc.org", "code": "53903-1", "display": "Collection method - Specimen" }] }, "entry": [{ "reference": "Specimen/spe-LABDM" }] }, { "code": { "coding": [{ "system": "http://loinc.org", "code": "11502-2", "display": "Laboratory report" }] }, "entry": [{ "reference": "Observation/ms-pmn" },{ "reference": "Observation/ms-ep" },{ "reference": "Observation/ac-p" },{ "reference": "Observation/detect" },{ "reference": "Observation/count" },{ "reference": "Observation/sus" },{ "reference": "DiagnosticReport/DR-gs" },{ "reference": "DiagnosticReport/DR-ac" }] }] }
{ "resourceType": "DiagnosticReport", "id":"diag-LABDM-DR-gs", "status":"final", "category": [{ "coding": [{ "system": "http://terminology.hl7.org/CodeSystem/v2-0074", "code": "LAB", "display": "Laboratory" }], "text": "Laboratory" }], "code": { "coding": [{ "system": "https://twcore.mohw.gov.tw/ig/twcore/CodeSystem/medical-service-payment-tw", "code": "13006C", "display": "排泄物,滲出物及分泌物之細菌顯微鏡檢查" }], "text": "顯微鏡檢查(使用革蘭氏染色)" }, "subject": { "reference": "Patient/pat-CDM" }, "result": [{ "reference": "Observation/obs-LABDM-ms-pmn" },{ "reference": "Observation/obs-LABDM-ms-ep" }], "conclusion": "0" }
{ "resourceType": "Observation", "id": "obs-LABDM-ms-pmn", "identifier": [{ "value": "1" }], "status": "final", "category": [{ "coding": [{ "system": "http://terminology.hl7.org/CodeSystem/observation-category", "code": "laboratory" }] }], "code": { "coding": [{ "system": "https://twcore.mohw.gov.tw/ig/twcore/CodeSystem/medical-service-payment-tw", "code": "13006C" },{ "system": "http://loinc.org", "code": "72163-9", "display": "Leukocytes [Presence] in Specimen by Gram stain" }], "text":"痰液中的Leukocyte是檢查polymorphonuclear cell,未來若有更適合的代碼會再修改" }, "subject": { "reference": "Patient/pat-CDM" }, "encounter": { "reference": "Encounter/enc-LABD" }, "effectiveDateTime": "2023-07-01T08:57:00+08:01", "method": { "coding": [{ "system": "http://snomed.info/sct", "code": "708061008" }], "text": "Gram stain" }, "valueString": ">25/100X", "referenceRange": [{ "text": "無資料" }] }
- 痰液中的Leukocyte是檢查polymorphonuclear cell。現行 LOINC 尚無對應之代碼,暫採「Leukocytes [Presence] in Specimen by Gram stain」。待後續釋出更合適之標準代碼時,將依標準更新。
{ "resourceType": "Observation", "id": "obs-LABDM-ms-ep", "identifier": [{ "value": "1" }], "status": "final", "category": [{ "coding": [{ "system": "http://terminology.hl7.org/CodeSystem/observation-category", "code": "laboratory" }] }], "code": { "coding": [{ "system": "https://twcore.mohw.gov.tw/ig/twcore/CodeSystem/medical-service-payment-tw", "code": "13006C" },{ "system": "http://loinc.org", "code": "72161-3", "display": "Epithelial cells [Presence] in Specimen by Gram stain" }], "text": "檢查EPITHELIUM" }, "subject": { "reference": "Patient/pat-CDM" }, "encounter": { "reference": "Encounter/enc-LABD" }, "effectiveDateTime": "2023-07-01T08:57:00+08:01", "method": { "coding": [{ "system": "http://snomed.info/sct", "code": "708061008" }], "text": "Gram stain" }, "valueString": "10-25/100x", "referenceRange": [{ "text":"無資料" }] }
{ "resourceType": "DiagnosticReport", "id": "diag-LABDM-DR-ac", "status": "final", "category": [{ "coding": [{ "system": "http://terminology.hl7.org/CodeSystem/v2-0074", "code": "LAB", "display": "Laboratory" }], "text": "Laboratory" }], "code": { "coding": [{ "system": "http://loinc.org", "code": "11502-2", "display": "Laboratory report" }], "text": "細菌培養鑑定與抗生素抗藥性檢查" }, "subject": { "reference": "Patient/pat-CDM" }, "result": [{ "reference": "Observation/obs-LABDM-ac-p" },{ "reference": "Observation/obs-LABDM-detect" },{ "reference": "Observation/obs-LABDM-count" },{ "reference": "Observation/obs-LABDM-sus" }], "conclusion": "0" }
{ "resourceType": "Observation", "id": "obs-LABDM-detect", "identifier": [{ "value": "1" }], "status": "final", "category": [{ "coding": [{ "system": "http://terminology.hl7.org/CodeSystem/observation-category", "code": "laboratory" }] }], "code": { "coding": [{ "system": "https://twcore.mohw.gov.tw/ig/twcore/CodeSystem/medical-service-payment-tw", "code": "13007C" },{ "system": "http://loinc.org", "code": "6463-4", "display": "Bacteria identified in Specimen by Culture" }], "text": "細菌鑑定(有氧培養)" }, "subject": { "reference": "Patient/pat-CDM" }, "encounter": { "reference": "Encounter/enc-LABD" }, "effectiveDateTime": "2023-07-01T08:57:00+08:01", "method": { "coding": [{ "system": "http://snomed.info/sct", "code": "7882003", "display": "Identified (qualifier value)" }], "text": "細菌鑑定" }, "valueCodeableConcept": { "coding": [{ "system": "http://snomed.info/sct", "code": "91288006", "display": "Acinetobacter baumannii (organism)" }], "text": "Acinetobacter baumannii" }, "referenceRange": [{ "text": "無資料" }] }
{ "resourceType": "Observation", "id": "obs-LABDM-ac-p", "identifier": [{ "value": "1" }], "status": "final", "category": [{ "coding": [{ "system": "http://terminology.hl7.org/CodeSystem/observation-category", "code": "laboratory" }] }], "code": { "coding": [{ "system": "https://twcore.mohw.gov.tw/ig/twcore/CodeSystem/medical-service-payment-tw", "code": "13007C" },{ "system": "http://loinc.org", "code": "LP6103-8", "display":"Aerobic Culture" }], "text": "有氧培養,待LOINC更新有適合的代碼時,將修改代碼" }, "subject": { "reference": "Patient/pat-CDM" }, "encounter": { "reference": "Encounter/enc-LABD" }, "effectiveDateTime": "2023-07-01T08:57:00+08:01", "method": { "coding": [{ "system": "http://snomed.info/sct", "code": "703750006", "display": "Aerobic Culture" }], "text": "有氧培養" }, "valueCodeableConcept": { "coding": [{ "system": "http://snomed.info/sct", "code": "10828004", "display": "Positive" }], "text": "Aerobic culture positive" }, "referenceRange": [{ "text": "無資料" }] }
本檔目前暫以LOINC Part code標示「有氧培養(Aerobic Culture)」。待 LOINC 更新並提供適用之標準代碼後,將依標準進行代碼調整與更新。
{ "resourceType": "Observation", "id": "obs-LABDM-count", "identifier": [{ "value": "1" }], "status": "final", "category": [{ "coding": [{ "system": "http://terminology.hl7.org/CodeSystem/observation-category", "code": "laboratory" }] }], "code": { "coding": [{ "system": "https://twcore.mohw.gov.tw/ig/twcore/CodeSystem/medical-service-payment-tw", "code": "13007C" },{ "system": "http://loinc.org", "code": "LP16817-6", "display": "Colony count" }], "text": "菌落計數" }, "subject": { "reference": "Patient/pat-CDM" }, "encounter": { "reference": "Encounter/enc-LABD" }, "effectiveDateTime": "2023-07-01T08:57:00+08:00", "method": { "coding": [{ "system": "http://snomed.info/sct", "code": "118022004", "display": "Colony count (procedure)" }], "text": "菌落計數" }, "valueCodeableConcept": { "coding": [{ "system": "http://snomed.info/sct", "code": "1255665007", "display": "Moderate" }], "text": "MODERATE" }, "referenceRange": [{ "text": "無資料" }] }
{ "resourceType": "Observation", "id": "obs-LABDM-sus", "identifier": [{ "value": "1" }], "status": "final", "category": [{ "coding": [{ "system": "http://terminology.hl7.org/CodeSystem/observation-category", "code": "laboratory" }] }], "code": { "coding": [{ "system": "https://twcore.mohw.gov.tw/ig/twcore/CodeSystem/medical-service-payment-tw", "code": "13023C" },{ "system": "http://loinc.org", "code": "29576-6", "display": "Bacterial susceptibility panel" }], "text": "藥物敏感性試驗" }, "subject": { "reference": "Patient/pat-CDM" }, "encounter": { "reference": "Encounter/enc-LABD" }, "effectiveDateTime": "2023-07-01T08:57:00+08:01", "method": { "coding": [{ "system": "http://snomed.info/sct", "code": "708073008", "display": "Minimum inhibitory concentration susceptibility test" }], "text": "細菌最低抑制濃度快速試驗" }, "component": [{ "code": { "coding": [{ "system": "http://loinc.org", "code": "28-1", "display": "Ampicillin [Susceptibility] by Minimum inhibitory concentration (MIC)" }] }, "valueQuantity": { "value": 32, "comparator": ">=" }, "interpretation": [{ "coding": [{ "system": "http://terminology.hl7.org/CodeSystem/v3-ObservationInterpretation", "code": "R", "display": "Resistant" }], "text": "RESISTANT" }] },{ "code": { "coding": [{ "system": "http://loinc.org", "code": "76-0", "display": "ceFAZolin [Susceptibility] by Minimum inhibitory concentration (MIC)" }] }, "valueQuantity": { "value": 64, "comparator": ">=" }, "interpretation": [{ "coding": [{ "system": "http://terminology.hl7.org/CodeSystem/v3-ObservationInterpretation", "code": "R", "display": "Resistant" }], "text": "RESISTANT" }] },{ "code": { "coding": [{ "system": "http://loinc.org", "code": "267-5", "display": "Gentamicin [Susceptibility] by Minimum inhibitory concentration (MIC)" }] }, "valueQuantity": { "value": 8 }, "interpretation": [{ "coding": [{ "system": "http://terminology.hl7.org/CodeSystem/v3-ObservationInterpretation", "code": "I", "display": "Intermediate" }], "text": "INTERMEDIATE" }] },{ "code": { "coding": [{ "system": "http://loinc.org", "code": "12-5", "display": "Amikacin [Susceptibility] by Minimum inhibitory concentration (MIC)" }] }, "valueQuantity": { "value": 2, "comparator": "<=" }, "interpretation": [{ "coding": [{ "system": "http://terminology.hl7.org/CodeSystem/v3-ObservationInterpretation", "code": "S", "display": "Susceptible" }], "text": "SUSCEPTIBLE" }] }], "referenceRange": [{ "text": "無資料" }] }